Diabetic nephropathy (DN) is one of the most crucial continuous kidney illnesses and the essential set off of end-stage renal failure worldwide. The underlying mechanisms of DN are superior and required to be further investigated. Both innate immunity and renin-angiotensin system (RAS) play very important roles in the pathogenesis of DN.
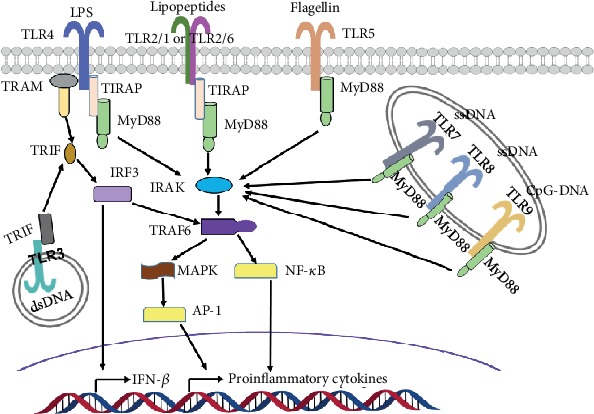
Except for standard capabilities, abnormally regulated RAS has been proved to be involved in the inflammatory course of of DN. Toll-like receptor 4 (TLR4) is the most deeply studied pattern recognition receptor in the innate immune system, and its activation has been reported to mediate the development of DN.
In this evaluation, we intention at discussing how dysregulated RAS impacts TLR4 activation in the kidney that contributes to the exploration of the pathogenesis of DN. Understanding the interplay of RAS and TLR4 in inducing the growth of DN may current new insights to develop environment friendly cures.
Triggering receptor expressed on myeloid Cells 2 activation downregulates toll-like receptor 4 expression and ameliorates cognitive impairment in the Aβ1-42 -induced Alzheimer’s sickness mouse model.
Increasing proof signifies that changes in the triggering receptor expressed on myeloid cells 2 (TREM2) is intently correlated with the pathological development of Alzheimer’s sickness (AD). However, the natural carry out and related place of this modification keep poorly understood. Higher TREM2 expression has been reported in the thoughts of AD victims than in common controls.
Here, ranges of TREM2 gene and protein ranges have been observed to be elevated in every cortex and hippocampus of the Aβ1-42 -induced AD mice than in these of the wild-type mice.
Together with in vitro experimental data, we found that the anti-inflammatory place of TREM2 was, to some extent, restricted and doubtlessly counteracted by the hyperactive toll-like receptor 4 (TLR4) in the AD mice. In this context, Interleukin 4 (IL-4), as an agonist of TREM2, was administered to the AD mice to persistently activate TREM2.
Interestingly, TREM2 activation in IL-4-treated AD mice led to an elevation in lysosomes and microtubule-associated protein 1 gentle chain 3 (LC3) II/I expression, demonstrating that the diploma of microglia autophagy was elevated. Increased autophagy significantly downregulated the expression ranges of caspase recruitment domain-containing protein 9 (CARD9) and TLR4, doubtlessly weakening the CARD9-TLR4 pathway and suppressing the TLR4-mediated pro-inflammatory influence in IL-4-treated AD mice. Furthermore, data acquired from Morris water maze (MWM) testing indicated that IL-4 administration may ameliorate cognitive impairment in the AD mice.
In conclusion, the findings from in vitro and in vivo experiments suggest that TREM2 could symbolize a attainable drug objective to take care of neuroinflammation in AD.